SN1 vs SN2 Reaction Practice Questions with Answers
Concept Explanation
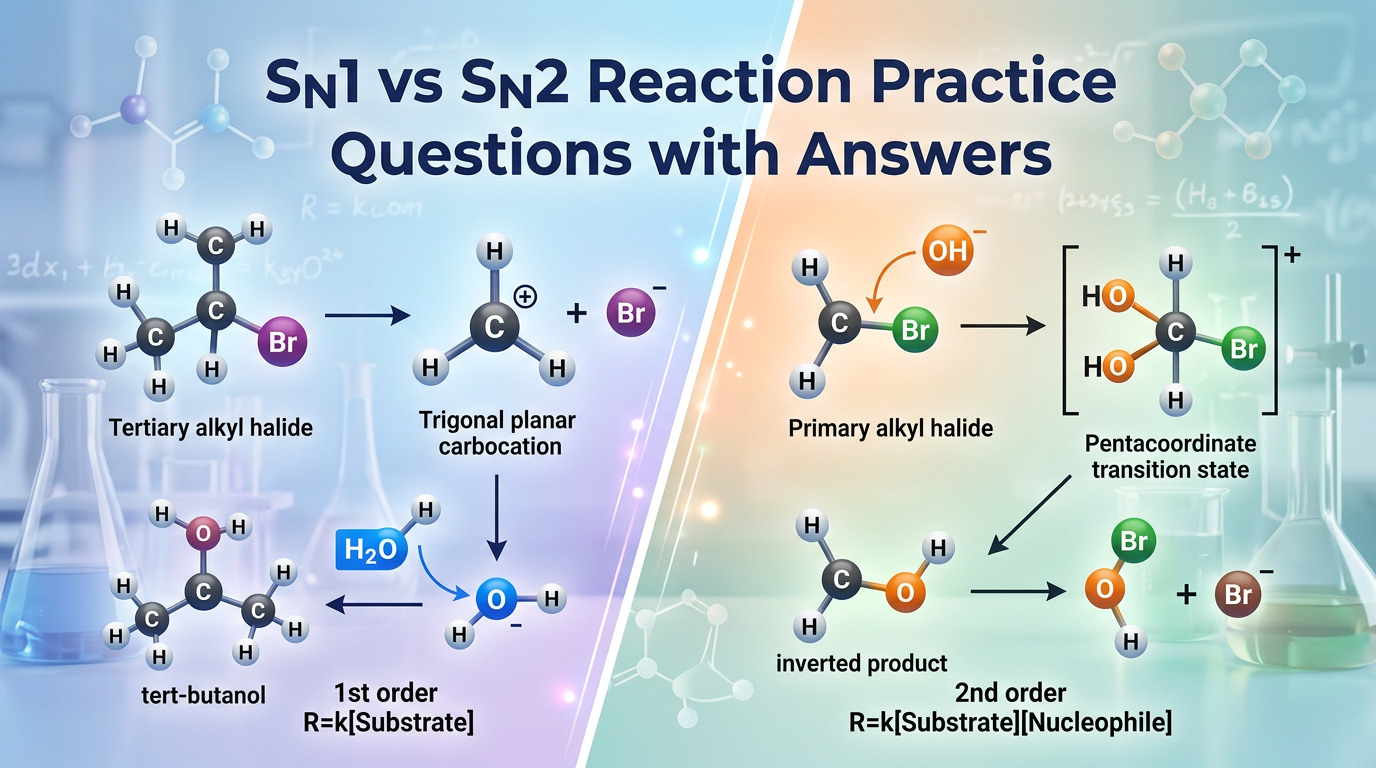
The SN1 and SN2 reaction pathways represent the two primary mechanisms of nucleophilic substitution where a nucleophile replaces a leaving group on a carbon atom. Understanding the SN1 vs SN2 reaction distinction is essential for predicting the outcome of organic transformations. The SN1 (Substitution Nucleophilic Unimolecular) mechanism occurs in two steps: first, the leaving group departs to form a carbocation intermediate, and second, the nucleophile attacks. Because the rate-determining step involves only the substrate, the reaction follows first-order kinetics. Conversely, the SN2 (Substitution Nucleophilic Bimolecular) mechanism is a concerted, one-step process where the nucleophile attacks the substrate at the same time the leaving group departs, leading to second-order kinetics and a complete inversion of stereochemistry at the reaction center.
Several factors dictate which pathway a reaction will follow. Substrate structure is the most influential; tertiary carbons favor SN1 due to the stability of the resulting carbocation, while primary carbons favor SN2 because they offer minimal steric hindrance to the incoming nucleophile. Secondary carbons are often the most difficult to predict and require looking at other factors like solvent and nucleophile strength. Strong nucleophiles (usually negatively charged) push a reaction toward SN2, whereas weak nucleophiles (often neutral like water or alcohols) are common in SN1. Furthermore, polar protic solvents stabilize carbocations and favor SN1, while polar aprotic solvents favor SN2 by increasing the reactivity of the nucleophile. For more on how molecular shapes influence reactivity, you might explore VSEPR geometry practice questions.
Feature SN1 Reaction SN2 Reaction Kinetics First order: Rate = k[Substrate] Second order: Rate = k[Substrate][Nu] Mechanism Two-step (via carbocation) One-step (concerted) Stereochemistry Racemization (loss of chirality) Inversion (Walden inversion) Substrate Preference 3° > 2° >> 1° CH3 > 1° > 2° >> 3°
Solved Examples
-
Example 1: Predicting the Mechanism
Determine if the reaction of 2-bromo-2-methylpropane with methanol proceeds via SN1 or SN2.-
Analyze the substrate: 2-bromo-2-methylpropane is a tertiary (3°) alkyl halide.
-
Analyze the nucleophile: Methanol (CH3OH) is a weak, neutral nucleophile.
-
Analyze the solvent: Methanol is a polar protic solvent.
-
Conclusion: Tertiary substrates cannot undergo SN2 due to steric hindrance, and weak nucleophiles in protic solvents favor the formation of a carbocation. This is an SN1 reaction.
-
-
Example 2: Stereochemical Outcome
Predict the product stereochemistry when (S)-2-iodobutane reacts with sodium cyanide (NaCN) in DMSO.-
Analyze the substrate: 2-iodobutane is a secondary (2°) alkyl halide.
-
Analyze the nucleophile: Cyanide (CN-) is a strong, negatively charged nucleophile.
-
Analyze the solvent: DMSO is a polar aprotic solvent, which accelerates SN2 reactions.
-
Conclusion: These conditions strongly favor SN2. The nucleophile attacks from the backside, resulting in an inversion of configuration. The product is (R)-2-cyanobutane.
-
-
Example 3: Rate Law Determination
If the concentration of the nucleophile is doubled in the reaction of methyl bromide with NaOH, what happens to the rate?-
Identify the substrate: Methyl bromide is a primary (methyl) halide, which exclusively follows the SN2 pathway.
-
Identify the rate law: For SN2, Rate = k[Substrate][Nucleophile].
-
Calculate the change: Since the rate is directly proportional to the nucleophile concentration, doubling [NaOH] will double the reaction rate.
-
Practice Questions
-
Which mechanism is favored when 1-chlorobutane reacts with sodium ethoxide in ethanol?
-
Rank the following in order of increasing SN2 reactivity: 2-bromo-2-methylbutane, 1-bromobutane, 2-bromobutane.
-
True or False: A polar aprotic solvent like acetone decreases the rate of an SN2 reaction compared to water.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →-
Explain why (R)-3-chloro-3-methylhexane becomes a racemic mixture when dissolved in water.
-
What is the effect on the rate of an SN1 reaction if the concentration of the substrate is halved?
-
A secondary alkyl halide reacts with a strong nucleophile in a polar aprotic solvent. Which mechanism dominates?
-
Why are tertiary alkyl halides essentially unreactive toward SN2 mechanisms?
-
Predict the major product and mechanism for the reaction of 1-iodo-1-methylcyclohexane with ethanol.
-
Which is a better leaving group for both SN1 and SN2: Fluoride (F-) or Iodide (I-)?
-
How does the stability of a carbocation relate to the rate of an SN1 reaction?
Answers & Explanations
-
SN2. 1-chlorobutane is a primary alkyl halide. Sodium ethoxide is a strong nucleophile. Primary substrates with strong nucleophiles strongly favor the concerted SN2 pathway.
-
2-bromo-2-methylbutane < 2-bromobutane < 1-bromobutane. SN2 reactivity is governed by steric hindrance. The tertiary halide is least reactive, followed by the secondary, with the primary halide being the most reactive.
-
False. Polar aprotic solvents actually increase the rate of SN2 reactions because they do not solvate the nucleophile strongly, leaving it more "naked" and reactive. This is a common topic in polarity determination studies.
-
SN1 Mechanism. The substrate is tertiary and the solvent (water) is a weak nucleophile/polar protic solvent. The reaction forms a planar carbocation intermediate. The nucleophile can attack from either side with equal probability, leading to a 50:50 mixture of (R) and (S) enantiomers (racemization).
-
The rate is halved. The SN1 rate law is Rate = k[Substrate]. Since the rate is directly proportional to the substrate concentration, halving the concentration results in halving the rate.
-
SN2. Secondary halides can go either way, but the combination of a strong nucleophile and a polar aprotic solvent (which favors SN2) will drive the reaction through the SN2 pathway.
-
Steric Hindrance. In an SN2 reaction, the nucleophile must attack the carbon from the backside (180° from the leaving group). In a tertiary halide, bulky methyl or alkyl groups block this approach, making the transition state energy prohibitively high.
-
Mechanism: SN1; Product: 1-ethoxy-1-methylcyclohexane. The substrate is tertiary, and ethanol is a weak nucleophile and protic solvent. These conditions favor the formation of a tertiary carbocation.
-
Iodide (I-). Iodide is a better leaving group because it is a weaker base (the conjugate base of the strong acid HI) and is more polarizable, which helps stabilize the negative charge as it leaves. This follows general leaving group trends found in IUPAC literature.
-
Directly proportional. The rate-determining step of an SN1 reaction is the formation of the carbocation. According to the Hammond Postulate, factors that stabilize the carbocation (like hyperconjugation or resonance) lower the activation energy, thereby increasing the reaction rate.
Quick Quiz
1. Which of the following substrates is most reactive in an SN2 reaction?
- A 2-chloro-2-methylpropane
- B 2-chloropropane
- C Chloromethane
- D 1-chlorobutane
Check answer
Answer: C. Chloromethane
2. What is the characteristic kinetic order of an SN1 reaction?
- A Zero-order
- B First-order
- C Second-order
- D Third-order
Check answer
Answer: B. First-order
3. Which solvent would best facilitate an SN2 reaction between NaOH and CH3I?
- A Water
- B Ethanol
- C DMSO
- D Acetic acid
- E
Check answer
Answer: C. DMSO
4. What happens to the configuration of a chiral center during a successful SN2 attack?
- A It is retained
- B It is inverted
- C It is racemized
- D It becomes achiral
Check answer
Answer: B. It is inverted
5. Which intermediate is formed during an SN1 reaction?
- A Carbanion
- B Free radical
- C Carbocation
- D Pentacoordinate transition state
Check answer
Answer: C. Carbocation
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
What is the main difference between SN1 and SN2?
The main difference lies in the timing of the bond-breaking and bond-forming steps. SN1 is a two-step process involving a carbocation intermediate, while SN2 is a single-step concerted process where the nucleophile attacks as the leaving group leaves.
Why does SN1 result in racemization?
SN1 results in racemization because the intermediate carbocation is sp2 hybridized and planar. The nucleophile can attack the empty p-orbital from either the top or bottom face with equal ease, creating a mixture of enantiomers.
How do I choose between SN1 and SN2 for a secondary alkyl halide?
For secondary halides, look at the nucleophile and solvent. Strong nucleophiles and polar aprotic solvents favor SN2, while weak nucleophiles and polar protic solvents favor SN1.
Why are polar aprotic solvents better for SN2?
Polar aprotic solvents do not have hydrogen atoms bonded to electronegative elements, so they cannot form hydrogen bonds with the nucleophile. This prevents the nucleophile from being "caged" by solvent molecules, keeping it highly reactive for the backside attack.
Does the strength of the nucleophile affect SN1 rates?
No, the strength of the nucleophile does not affect the rate of an SN1 reaction. The rate-determining step only involves the dissociation of the substrate to form a carbocation, which occurs before the nucleophile participates.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.
Related Articles
Newton’s Laws Practice Questions with Answers
Master Newton's Laws with our comprehensive guide. Features clear explanations, solved examples, practice questions with answers, a quick quiz, and FAQs.
Apr 7, 2026

Velocity and Acceleration Practice Questions with Answers
Master the core physics concepts of motion with our comprehensive guide to velocity and acceleration. Includes solved examples, practice questions, and detailed answers.
Apr 7, 2026
Circuit Practice Questions with Answers
Sharpen your understanding of electrical circuits with practice questions. This guide covers Ohm's Law, series circuits, and parallel circuits with detailed examples.
Apr 7, 2026