Reaction Quotient (Q) Practice Questions with Answers
Concept Explanation
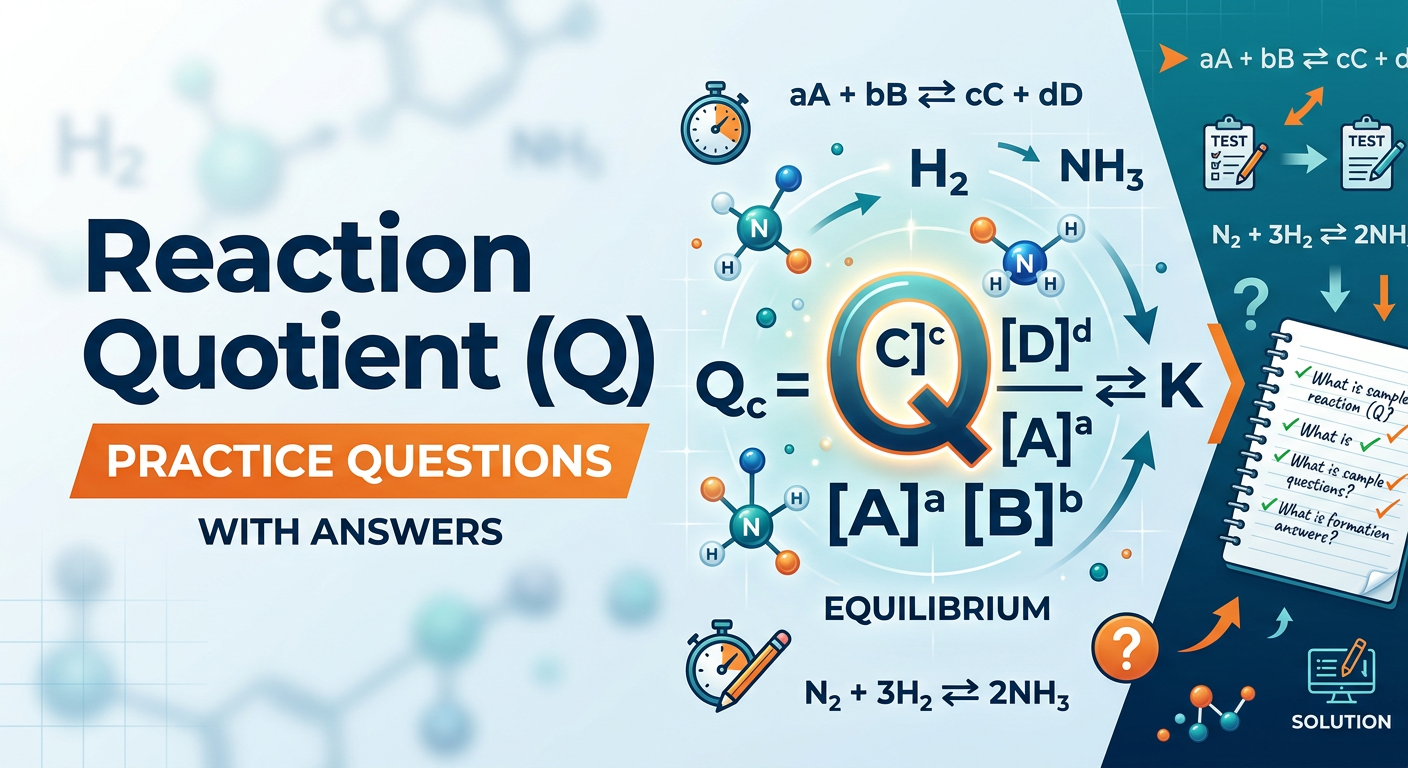
The Reaction Quotient (Q) is a mathematical expression that uses the current concentrations or partial pressures of reactants and products to determine the direction in which a reversible chemical reaction will proceed to reach equilibrium. While the Equilibrium Constant (K) describes the system at a state of balance, the reaction quotient can be calculated at any point in time, whether the system is at equilibrium or not. By comparing Q to K, chemists can predict if a reaction will shift toward the products (forward) or toward the reactants (reverse). For a generalized reaction aA + bB ⇌ cC + dD, the expression is written as Q = [C]c[D]d / [A]a[B]b.
Understanding the relationship between Q and K is critical for mastering chemical kinetics and thermodynamics. This concept is closely related to other energetic calculations, such as those found in Heat of Reaction Practice Questions with Answers. The three possible scenarios when comparing these values are:
-
Q < K: The ratio of products to reactants is less than the equilibrium ratio. The reaction will proceed in the forward direction (to the right) to form more products.
-
Q > K: The ratio of products to reactants is greater than the equilibrium ratio. The reaction will proceed in the reverse direction (to the left) to form more reactants.
-
Q = K: The system is at chemical equilibrium, and no net change in concentrations will occur.
It is important to remember that pure solids and liquids are excluded from the Q expression, as their activities are defined as 1. This rule is a staple in general chemistry courses, such as those offered by Khan Academy or documented in Wikipedia's entry on Reaction Quotients. For more advanced thermodynamic context, you might also explore Hard Enthalpy Change Practice Questions to see how energy and equilibrium interrelate.
Solved Examples
Example 1: Calculating Q for a Gas Phase Reaction
For the reaction: N2(g) + 3H2(g) ⇌ 2NH3(g), Kc = 0.50 at 400°C. If a container holds [N2] = 0.10 M, [H2] = 0.30 M, and [NH3] = 0.02 M, determine the direction of the reaction.
-
Write the Q expression: Qc = [NH3]2 / ([N2][H2]3).
-
Substitute the given values: Qc = (0.02)2 / (0.10 × 0.303).
-
Calculate the result: Qc = 0.0004 / (0.10 × 0.027) = 0.0004 / 0.0027 ≈ 0.148.
-
Compare Q to K: Since Q (0.148) < K (0.50), the reaction will shift to the right (forward) to reach equilibrium.
Example 2: Heterogeneous Equilibrium
Consider the decomposition of calcium carbonate: CaCO3(s) ⇌ CaO(s) + CO2(g). At a certain temperature, Kp = 1.2. If the partial pressure of CO2 is 2.5 atm, which way will the reaction shift?
-
Identify the species for the Q expression: Since CaCO3 and CaO are solids, they are omitted.
-
Write the expression: Qp = PCO2.
-
Plug in the value: Qp = 2.5.
-
Compare: Q (2.5) > K (1.2). The reaction will shift to the left (reverse), forming more solid CaCO3.
Example 3: Determining Equilibrium State
For the reaction H2(g) + I2(g) ⇌ 2HI(g), Kc = 50.0. A 2.0 L flask contains 1.0 mol H2, 1.0 mol I2, and 7.07 mol HI. Is the system at equilibrium?
-
Calculate molar concentrations: [H2] = 1.0/2.0 = 0.5 M; [I2] = 1.0/2.0 = 0.5 M; [HI] = 7.07/2.0 = 3.535 M.
-
Write the Q expression: Qc = [HI]2 / ([H2][I2]).
-
Substitute: Qc = (3.535)2 / (0.5 × 0.5) = 12.496 / 0.25 = 49.98.
-
Compare: Q ≈ 50.0. Since Q = K, the system is at equilibrium.
Practice Questions
1. For the reaction 2SO2(g) + O2(g) ⇌ 2SO3(g), Kc = 100. If [SO2] = 0.1 M, [O2] = 0.1 M, and [SO3] = 1.0 M, calculate Q and predict the shift direction.
2. A mixture contains 0.5 mol of A and 0.5 mol of B in a 1.0 L container for the reaction A(g) ⇌ B(g) where K = 2.0. In which direction must the reaction proceed?
3. Consider the reaction PCl5(g) ⇌ PCl3(g) + Cl2(g) with Kc = 0.040. If the concentrations are [PCl5] = 0.50 M, [PCl3] = 0.20 M, and [Cl2] = 0.20 M, determine if the system is at equilibrium.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →4. For the reaction 2NO(g) + Br2(g) ⇌ 2NOBr(g), Kc = 1.3. If the initial concentrations are [NO] = 0.2 M, [Br2] = 0.5 M, and [NOBr] = 0.1 M, calculate Q.
5. In the reaction C(s) + H2O(g) ⇌ CO(g) + H2(g), Kp = 2.0. If PH2O = 0.5 atm, PCO = 2.0 atm, and PH2 = 2.0 atm, which way will the reaction shift?
6. The reaction 2H2S(g) ⇌ 2H2(g) + S2(g) has Kc = 1.0 × 10-6. If a flask contains 0.10 M H2S, 1.0 × 10-3 M H2, and 1.0 × 10-3 M S2, calculate Q and compare it to K.
7. For the reaction N2O4(g) ⇌ 2NO2(g), Kc = 4.6 × 10-3. If [N2O4] = 0.20 M and [NO2] = 0.01 M, determine the direction of the reaction.
8. Liquid water and water vapor are in equilibrium: H2O(l) ⇌ H2O(g). At 25°C, Kp = 0.0313 atm. If the partial pressure of water vapor is 0.050 atm, what happens to the amount of liquid water?
9. A reaction A + 2B ⇌ C has Kc = 10. If [A] = 2.0 M, [B] = 1.0 M, and [C] = 5.0 M, is the reaction moving toward products or reactants?
10. For the synthesis of methanol, CO(g) + 2H2(g) ⇌ CH3OH(g), Kc = 10.5. At a specific moment, [CO] = 0.5 M, [H2] = 0.5 M, and [CH3OH] = 0.2 M. Calculate Q and predict the shift.
Answers & Explanations
-
Q = 1000; Shift Left. Q = [SO3]2 / ([SO2]2[O2]) = (1.0)2 / ((0.1)2 × 0.1) = 1 / 0.001 = 1000. Since Q (1000) > K (100), the reaction shifts left.
-
Shift Right. Q = [B]/[A] = 0.5/0.5 = 1.0. Since Q (1.0) < K (2.0), the reaction shifts right to produce more B.
-
Shift Left. Q = ([PCl3][Cl2]) / [PCl5] = (0.2 × 0.2) / 0.5 = 0.04 / 0.5 = 0.08. Since Q (0.08) > K (0.04), the system is not at equilibrium and shifts left.
-
Q = 0.5. Q = [NOBr]2 / ([NO]2[Br2]) = (0.1)2 / ((0.2)2 × 0.5) = 0.01 / (0.04 × 0.5) = 0.01 / 0.02 = 0.5.
-
Shift Left. Qp = (PCO × PH2) / PH2O = (2.0 × 2.0) / 0.5 = 4 / 0.5 = 8.0. Since Q (8.0) > K (2.0), the reaction shifts left. Note that C(s) is ignored.
-
Q = 1.0 × 10-4; Shift Left. Q = ([H2]2[S2]) / [H2S]2 = ((10-3)2 × 10-3) / (0.1)2 = 10-9 / 0.01 = 10-7. Wait, let's re-calculate: (10-6 × 10-3) / 0.01 = 10-9 / 0.01 = 10-7. Since Q (10-7) < K (10-6), it actually shifts Right. (Self-correction: 10-7 is smaller than 10-6).
-
Shift Right. Q = [NO2]2 / [N2O4] = (0.01)2 / 0.20 = 0.0001 / 0.20 = 0.0005. Since Q (0.0005) < K (0.0046), the reaction shifts right.
-
Increases. Qp = PH2O = 0.050. Since Q (0.050) > K (0.0313), the reaction shifts left toward H2O(l), increasing the amount of liquid.
-
Shift Left. Q = [C] / ([A][B]2) = 5.0 / (2.0 × 1.02) = 5.0 / 2.0 = 2.5. Wait, K is 10. Since Q (2.5) < K (10), the reaction shifts Right.
-
Q = 1.6; Shift Right. Q = [CH3OH] / ([CO][H2]2) = 0.2 / (0.5 × 0.52) = 0.2 / (0.5 × 0.25) = 0.2 / 0.125 = 1.6. Since Q (1.6) < K (10.5), it shifts right.
Quick Quiz
1. What does it mean if the Reaction Quotient (Q) is equal to the Equilibrium Constant (K)?
- A The reaction is shifting toward products
- B The reaction is shifting toward reactants
- C The system is at equilibrium
- D The reaction has stopped completely
Check answer
Answer: C. The system is at equilibrium
2. If Q > K, in which direction will the reaction proceed to reach equilibrium?
- A Forward (to the right)
- B Reverse (to the left)
- C No shift will occur
- D The reaction will explode
Check answer
Answer: B. Reverse (to the left)
3. Which of the following states of matter are excluded from the Reaction Quotient expression?
- A Gases and aqueous solutions
- B Pure liquids and pure solids
- C Only pure solids
- D Only aqueous solutions
Check answer
Answer: B. Pure liquids and pure solids
4. For the reaction A + B ⇌ C, if you double the initial concentration of A, what happens to the initial value of Q?
- A Q doubles
- B Q stays the same
- C Q is halved
- D Q quadruples
Check answer
Answer: C. Q is halved
5. A reaction has K = 5.0. If the calculated Q is 2.0, what is the status of the reaction?
- A It is at equilibrium
- B It must form more reactants
- C It must form more products
- D The K value is incorrect
Check answer
Answer: C. It must form more products
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
What is the difference between Q and K?
Q represents the ratio of products to reactants at any given moment during a reaction, whereas K represents that same ratio specifically when the system has reached chemical equilibrium at a constant temperature.
Can the Reaction Quotient be negative?
No, the Reaction Quotient cannot be negative because it is calculated using concentrations or partial pressures, which are always zero or positive values.
How do you use Q to predict precipitation?
In solubility equilibria, Q (often called the ion product) is compared to the solubility product constant (Ksp); if Q > Ksp, the solution is supersaturated and a precipitate will form.
Why are solids omitted from the Q expression?
Solids are omitted because their concentration (density) remains constant throughout the reaction, and their thermodynamic activity is defined as unity (1) in the equilibrium expression.
Does temperature change the value of Q?
Temperature does not change the formula for Q, but it does change the value of the Equilibrium Constant (K), which alters the target Q must reach to achieve equilibrium.
Is Q related to the pH of a solution?
Yes, for acid-base reactions, Q can be calculated using the concentration of hydronium ions, which is directly related to pH as discussed in Easy pH Calculation Practice Questions.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.
Related Articles
Newton’s Laws Practice Questions with Answers
Master Newton's Laws with our comprehensive guide. Features clear explanations, solved examples, practice questions with answers, a quick quiz, and FAQs.
Apr 7, 2026

Velocity and Acceleration Practice Questions with Answers
Master the core physics concepts of motion with our comprehensive guide to velocity and acceleration. Includes solved examples, practice questions, and detailed answers.
Apr 7, 2026
Circuit Practice Questions with Answers
Sharpen your understanding of electrical circuits with practice questions. This guide covers Ohm's Law, series circuits, and parallel circuits with detailed examples.
Apr 7, 2026