Quantum Number Practice Questions with Answers
Concept Explanation
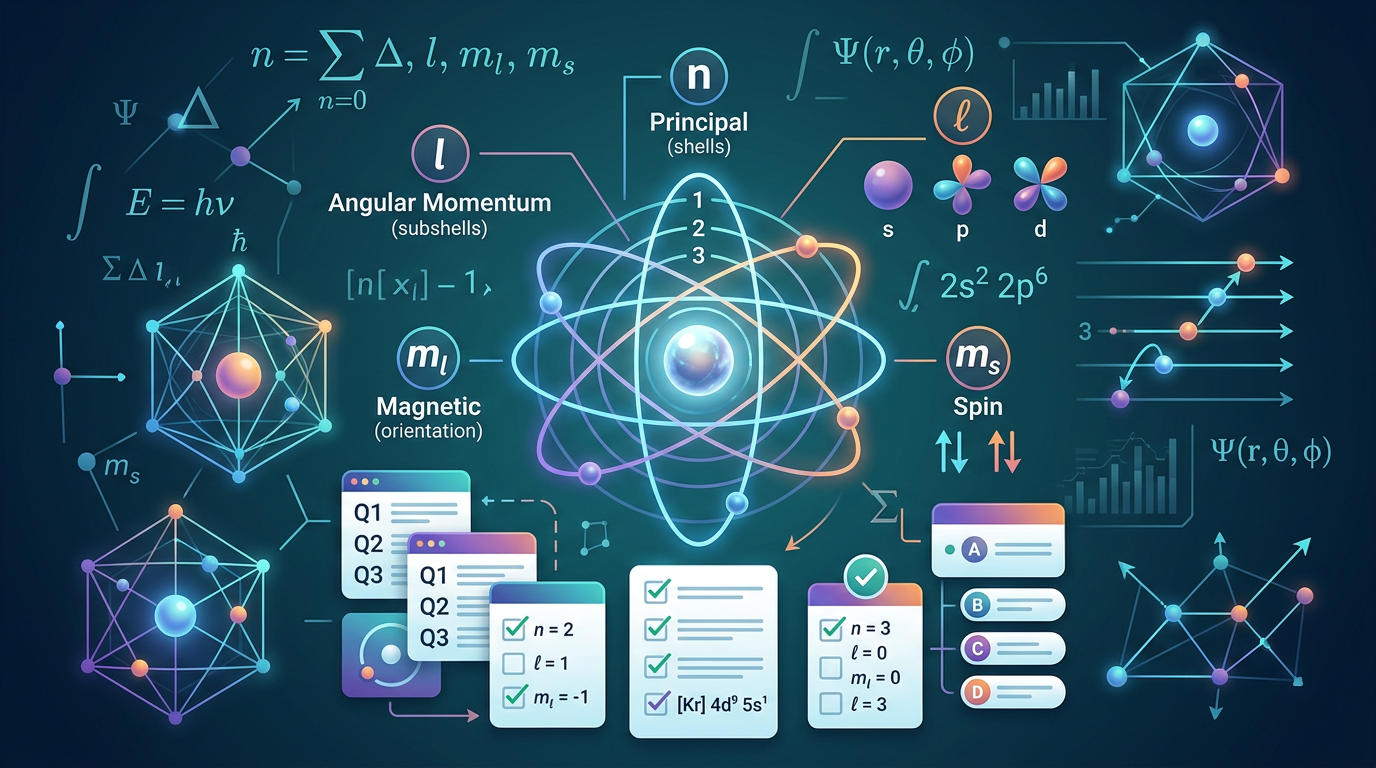
A quantum number is one of four values used to describe the unique energy state, spatial distribution, and orientation of an electron within an atom. According to the Schrödinger equation, these numbers act like a "postal address" for an electron, ensuring that no two electrons in a single atom share the exact same set of values—a principle known as the Pauli Exclusion Principle. Understanding how these numbers interact is essential for mastering chemical bonding and reactivity, much like how understanding the Ideal Gas Law is fundamental to gas stoichiometry.
The four quantum numbers are defined as follows:
-
Principal Quantum Number (n): Indicates the main energy level or shell. It must be a positive integer (n = 1, 2, 3...). As n increases, the electron's distance from the nucleus and its energy increase.
-
Angular Momentum Quantum Number (l): Defines the shape of the orbital (subshell). Its value ranges from 0 to (n - 1). Common values include 0 (s-orbital), 1 (p-orbital), 2 (d-orbital), and 3 (f-orbital).
-
Magnetic Quantum Number (ml): Describes the orientation of the orbital in space. Its values range from -l to +l, including zero.
-
Spin Quantum Number (ms): Describes the direction of the electron's intrinsic spin. It can only be +1/2 (spin up) or -1/2 (spin down).
When solving quantum number practice questions, you must ensure the values follow these mathematical constraints. For instance, if n=2, l can only be 0 or 1. If l=1, ml can only be -1, 0, or 1. This hierarchy governs the structure of the periodic table and the electron configuration of elements. Mastery of these concepts often precedes more advanced topics like Redox Reaction Practice Questions in general chemistry curricula.
Solved Examples
Below are worked examples demonstrating how to determine valid sets of quantum numbers and identify specific orbitals.
-
Identifying Orbital Type: What type of orbital is designated by the quantum numbers n = 4 and l = 2?
-
Identify the principal shell: n = 4, so it is the 4th shell.
-
Identify the subshell shape: l = 2 corresponds to a "d" orbital.
-
Result: The orbital is a 4d orbital.
-
-
Validating Quantum Sets: Is the set {n=2, l=2, ml=0, ms=+1/2} allowed? Explain why or why not.
-
Check the relationship between n and l: l must be less than n (l < n).
-
In this set, n=2 and l=2. Since l is not less than n, this set is invalid.
-
Result: Not allowed; for n=2, the maximum value for l is 1.
-
-
Calculating Total Orbitals: How many total orbitals are available in the n = 3 shell?
-
The total number of orbitals in a shell is calculated as n².
-
Calculate 3² = 9.
-
Breakdown: One 3s orbital (l=0), three 3p orbitals (l=1), and five 3d orbitals (l=2). 1 + 3 + 5 = 9.
-
Result: 9 orbitals.
-
Practice Questions
-
What are the possible values for the angular momentum quantum number (l) when the principal quantum number (n) is 4?
-
Identify the subshell label (e.g., 2p, 3s) for the following set: n = 5, l = 3.
-
Determine if the following set is valid: {n=3, l=0, ml=-1, ms=-1/2}. If not, explain why.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →-
How many electrons can be held in the 4f subshell?
-
What is the maximum value of ml for an electron in a 3d orbital?
-
Which quantum number determines the orientation of an orbital in space?
-
A specific electron has the quantum numbers {n=2, l=1, ml=0}. What is the name of the orbital this electron occupies?
-
If n=1, what are the only allowed values for l and ml?
-
How many distinct orbitals exist for the l=2 subshell?
-
Identify the error in this set: {n=4, l=3, ml=2, ms=0}.
Answers & Explanations
-
1. Answer: 0, 1, 2, 3. Since l ranges from 0 to (n-1), for n=4, l can be 0, 1, 2, or 3.
-
2. Answer: 5f. The principal number n=5 gives the shell, and l=3 corresponds to the f-subshell.
-
3. Answer: Invalid. When l=0 (an s-orbital), the only allowed value for ml is 0. Here, ml=-1 is impossible.
-
4. Answer: 14 electrons. The "f" subshell (l=3) has 7 orbitals (ml = -3, -2, -1, 0, 1, 2, 3). Each orbital holds 2 electrons, so 7 × 2 = 14.
-
5. Answer: +2. For a 3d orbital, l=2. The range of ml is -l to +l, so the maximum value is +2.
-
6. Answer: Magnetic Quantum Number (ml). While n determines size and l determines shape, ml defines the spatial orientation.
-
7. Answer: 2p orbital. n=2 and l=1 (which is the p-subshell) combined results in the 2p label.
-
8. Answer: l=0 and ml=0. When n=1, l can only be (1-1)=0. Because l=0, ml must also be 0.
-
9. Answer: 5 orbitals. The number of orbitals in a subshell is (2l + 1). For l=2, (2*2 + 1) = 5.
-
10. Answer: ms=0. The spin quantum number can only be +1/2 or -1/2. It cannot be 0.
Quick Quiz
1. Which quantum number defines the shape of an orbital?
- A Principal Quantum Number (n)
- B Angular Momentum Quantum Number (l)
- C Magnetic Quantum Number (ml)
- D Spin Quantum Number (ms)
Check answer
Answer: B. Angular Momentum Quantum Number (l)
2. What is the maximum number of electrons that can occupy the n=2 shell?
- A 2
- B 4
- C 8
- D 18
Check answer
Answer: C. 8
3. If l = 1, how many possible values are there for the magnetic quantum number (ml)?
- A 1
- B 2
- C 3
- D 4
Check answer
Answer: C. 3
4. Which of the following is NOT an allowed value for the angular momentum quantum number (l) when n = 3?
- A 0
- B 1
- C 2
- D 3
Check answer
Answer: D. 3
5. Which value represents a spin-down electron?
- A +1
- B -1/2
- C 0
- D +1/2
Check answer
Answer: B. -1/2
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
What is the physical meaning of the principal quantum number?
The principal quantum number (n) represents the relative size and energy level of an atomic orbital. As n increases, the electron spends more time further from the nucleus, and the orbital becomes larger and higher in energy.
Can two electrons in the same orbital have the same spin?
No, according to the Pauli Exclusion Principle, two electrons in the same orbital must have opposite spins. This means one must have ms = +1/2 and the other must have ms = -1/2.
How do I find the number of subshells in a given energy level?
The number of subshells in any principal energy level is equal to the value of the principal quantum number (n). For example, the n=3 shell contains three subshells: s, p, and d.
Why can the angular momentum quantum number (l) never be equal to n?
The mathematical solutions to the wave equation for an atom restrict the values of l to integers from 0 up to (n-1). If l equaled n, the kinetic energy component of the electron's motion would violate the stability requirements of the atomic model.
What is the difference between an orbital and a subshell?
A subshell is a group of orbitals with the same shape (same l value), such as the 2p subshell. An orbital is a specific region within that subshell (specific ml value) that can hold a maximum of two electrons.
How do quantum numbers relate to electron configuration?
Quantum numbers provide the rules for filling electron shells, such as the Aufbau principle and Hund's rule. They dictate the order and capacity of orbitals, which is why we fill 4s before 3d, a concept often explored in Reaction Order Practice Questions when discussing transition metal catalysis.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.
Related Articles
Newton’s Laws Practice Questions with Answers
Master Newton's Laws with our comprehensive guide. Features clear explanations, solved examples, practice questions with answers, a quick quiz, and FAQs.
Apr 7, 2026

Velocity and Acceleration Practice Questions with Answers
Master the core physics concepts of motion with our comprehensive guide to velocity and acceleration. Includes solved examples, practice questions, and detailed answers.
Apr 7, 2026
Circuit Practice Questions with Answers
Sharpen your understanding of electrical circuits with practice questions. This guide covers Ohm's Law, series circuits, and parallel circuits with detailed examples.
Apr 7, 2026